Fluorescence Lifetime Imaging Microscopy (FLIM)
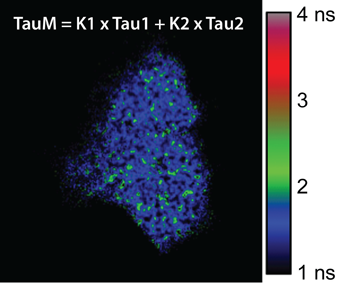
FLIM is a technique which measures the fluorescence lifetime of molecules. This is defined as the average time a molecule spends in an excited state before returning to the ground state. It is usually used to determine molecular dynamics with nanoscale resolution, predominately used with fluorescent molecules in cells, tissues, and whole animal models.
Fluorescence lifetime is dependent on changes within its local environment and conformation state, for example ionic concentration, pH, and lipophilicity. There are two primary implementations of FLIM:
- Time-domain FLIM; pulsed light source in conjunction with time-gated/time-correlated detection
- Frequency-domain FLIM – sinusoidally modulated light source with either a sinusoidally modulated ICCD for wide-field imaging or a modulated single-element detector for scanning/single-point detection.
Cameras for Fluorescence Lifetime Imaging Microscopy

PI-MAX4
Fluorescence lifetime imaging microscopy relies on pulsed illumination to determine fluorescence lifetimes. When a fluorophore is excited by a light pulse it decays exponentially. Therefore, a fast, reliable gating mechanism is required. The PI-MAX4 offers precision gating to <500 picoseconds, ideal for fluorescence decay.
The PI-MAX4 offers high quantum efficiency for fluorophores decaying in the wavelength region of 200 – 900 nm. With high precision timing and sensitivity, the ICCD sensor can capture most low-light.
The coupling of the EMCCD to an image intensifier allows for a 6x higher light throughput between the image intensifier and the detector in comparison to lens-coupled configurations. This ensures imaging of even the faintest of fluorophores over the UV-VIS-IR range.
